

 All payments are encrypted via SSL
All payments are encrypted via SSL
 Full Refund if you haven't received your order
Full Refund if you haven't received your order
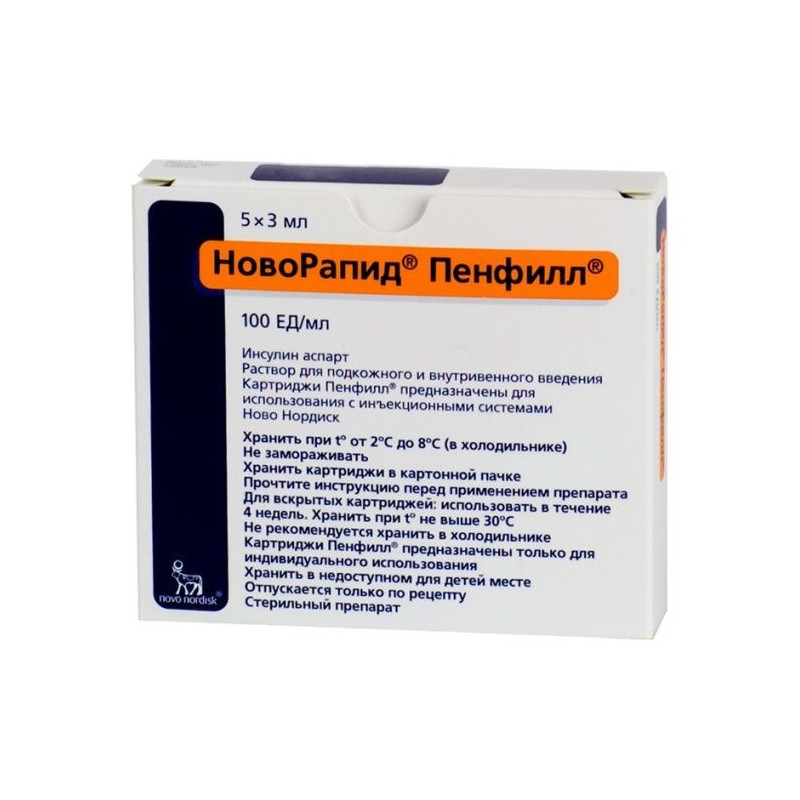
Solution for s / c and / in the introduction transparent, colorless.
1 ml | |
insulin aspart | 100 U * |
Excipients: glycerol - 16 mg, phenol - 1.5 mg, metacresol - 1.72 mg, zinc chloride - 19.6 μg, sodium chloride - 0.58 mg, sodium hydrogen phosphate dihydrate - 1.25 mg, sodium hydroxide 2M - about 2.2 mg, hydrochloric acid 2M - about 1.7 mg, water d / and - up to 1 ml.
3 ml - glass cartridges (5) - blisters (1) - cardboard packs.
* 1 U corresponds to 35 μg of anhydrous insulin aspart.
- diabetes in adults, adolescents and children over 2 years.
NovoRapid® Penfill® is a fast acting insulin analog. Dose NovoRapid ® Penfill® determined by the doctor individually in accordance with the needs of the patient.
Typically, the drug is used in combination with insulin preparations of medium duration or long-acting, which is administered at least 1 time / day. To achieve optimal glycemic control, it is recommended that you regularly measure your blood glucose concentration and adjust your insulin dose. Typically, individual daily insulin requirements in adults and children range from 0.5 to 1 U / kg body weight. With the introduction of the drug before a meal, the need for insulin can be provided by the drug NovoRapid® Penfill® 50-70%, the remaining need for insulin is provided by insulin with a prolonged action.
An increase in the patient's physical activity, a change in dietary habits, or concomitant diseases may lead to the need for dose adjustment.
NovoRapid® Penfill® has a faster onset and shorter duration of action than soluble human insulin. Thanks to a faster onset of action, NovoRapid® Penfill® should be administered, as a rule, immediately before a meal, if necessary, you can enter soon after a meal. Due to the shorter duration of action compared with human insulin, the risk of developing nighttime hypoglycemia in patients receiving NovoRapid® Penfill®below.
As with the use of other insulin preparations, elderly patients and patients with renal or hepatic insufficiency should be more closely control the concentration of glucose in the blood and adjust the dose of insulin aspart individually.
Children and teenagers. Apply NovoRapid® Penfill® instead of soluble human insulin in children, it is preferable when a rapid onset of action of the drug is necessary, for example, when it is difficult for the child to observe the necessary time interval between injection and food intake.
Transfer from other insulin preparations
When transferring a patient from other insulin preparations to NovoRapid® Penfill®, may require dose adjustment of the drug NovoRapid® Penfill® and basal insulin.
NovoRapid® Penfill® n / a in the area of the anterior abdominal wall, thigh, shoulder, deltoid or gluteal region. Injection sites within the same area of the body should be changed regularly to reduce the risk of developing lipodystrophy. As with the use of all insulin preparations, sc injection into the anterior abdominal wall provides a faster absorption compared with administration to other places. The duration of action depends on the dose, place of administration, intensity of blood flow, temperature and level of physical activity. However, a more rapid onset of action compared with soluble human insulin is maintained regardless of the location of the injection site.
NovoRapid® can be used for long-lasting insulin infusions (PPII) in insulin pumps designed for insulin infusions. FDII should be made in the front in the abdominal wall. Infusion sites should be periodically changed.
When using the insulin pump for infusions, NovoRapid® do not mix with other types of insulin.
Patients using PPII should be fully trained in the use of a pump, an appropriate reservoir and a tubing system for the pump. The infusion set (tube and catheter) should be replaced in accordance with the user's manual attached to the infusion set. Patients receiving NovoRapid® using the PPII should have additional insulin available in case of breakdown of the infusion system.
In / in the introduction
If necessary, NovoRapid® can be administered in / in, but only by qualified medical personnel.
For IV injection, NovoRapid infusion systems are used.® 100 U / ml with a concentration of 0.05 U / ml to 1 U / ml of insulin aspart in 0.9% sodium chloride solution, 5% dextrose solution or 10% dextrose solution containing 40 mmol / l potassium chloride, using polypropylene infusion containers. These solutions are stable at room temperature for 24 hours. Despite stability for some time, a certain amount of insulin is initially absorbed by the material of the infusion system. During insulin infusions it is necessary to constantly monitor the concentration of blood glucose.
Precautionary measures
NovoRapid® Penfill® and needles are intended for individual use only. Penfill cartridge refilling is not allowed.®.
NovoRapid® Penfill® cannot be used if it has ceased to be transparent and colorless, or if it has been frozen. Inform the patient of the need to throw out the needle after each injection.
NovoRapid® can be used in insulin pumps. Tubes, the inner surface of which is made of polyethylene or polyolefin, were checked and found to be suitable for use in pumps.
In case of emergency (hospitalization, malfunction of the device for insulin administration) NovoRapid® for administration to the patient can be removed from the cartridge using an U100 insulin syringe.
Instructions for patients on the use of NovoRapid® Penfill®
Do not use NovoRapid® Penfill®
- in case of allergy (hypersensitivity) to insulin aspart or any other component of NovoRapid®;
- if you start hypoglycemia (low blood glucose);
- if the cartridge or system for administering insulin with the cartridge installed is dropped, or the cartridge is damaged or crushed;
- if the storage conditions of the preparation were violated or it was frozen.;
- if insulin has ceased to be transparent and colorless.
Before using NovoRapid® Penfill®
- check the label to ensure that the correct type of insulin is selected;
- always check the cartridge, including the rubber piston. Do not use the cartridge if it is visibly damaged, or there is a gap between the piston and the white stripe on the cartridge.
For further guidance, refer to the instructions for use of the system for insulin administration.
- always use a new needle for each injection to prevent infection;
- NovoRapid® Penfill® and needles are intended for individual use only.
Mode of application
NovoRapid® intended for subcutaneous injections or continuous infusions in an insulin pumping system (PPII). NovoRapid® It can also be administered intravenously under strict medical supervision. Never inject insulin intramuscularly.
Each time, change the injection site within the anatomical region. This will help reduce the risk of seals and ulceration at the injection site. It is best to inject the drug into the anterior abdominal wall, shoulder, or front surface of the thigh. Insulin will act faster if it is injected into the anterior abdominal wall. Regularly measure your blood glucose concentration.
How to make an injection
Insulin should be administered under the skin. Use the injection technique recommended by your doctor or nurse, follow the instructions for administering insulin given in the manual for your insulin device.
Hold the needle under the skin for at least 6 seconds. Keep the trigger button pressed until the needle is removed from under the skin. This will ensure the introduction of a full dose of the drug and prevent blood from entering the needle or the cartridge with insulin.
After each injection, be sure to remove and throw away the needle. Otherwise, liquid may leak out of the cartridge, which may result in improper dosing of insulin.
Do not refill the cartridge with insulin. NovoRapid® Penfill® designed for use with Novo Nordisk injection systems for injection and NovoFine needles® or NovoTvist®.
If the drug NovoRapid is used simultaneously® Penfill® and another insulin in penfill cartridge®, you must use two separate injection systems for administering insulin, one for each type of insulin.As a precautionary measure, always carry with you a backup system for administering insulin in case of loss or damage to your NovoRapid® Penfill®.
Side effect
Adverse reactions observed in patients using the drug NovoRapid® Penfill®, mainly due to the pharmacological effect of insulin.
The incidence of side effects varies depending on the patient population, the dosage regimen of the drug and the control of glycemia (see section below). At the initial stage of insulin therapy, refractive disorders, edema and reactions in the places of drug administration (pain, redness, urticaria, inflammation, hematoma, swelling and itching at the injection site) can occur. These symptoms are usually transient. A rapid improvement in glycemic control can lead to a state of acute pain neuropathy, which is usually reversible.
Intensification of insulin therapy with a dramatic improvement in carbohydrate metabolism can lead to a temporary deterioration of diabetic retinopathy, while long-term improvement in glycemic control reduces the risk of progression of insulin therapy with a dramatic improvement in carbohydrate metabolism can lead to a temporary deterioration of diabetic retinopathy, while long-term improvement Glycemic control reduces the risk of progression of diabetic retinopathy.
A list of adverse reactions is presented in the table.
All of the adverse reactions presented below, based on data from clinical trials, are grouped according to the frequency of development according to MedDRA and organ systems. The incidence of adverse reactions is defined as follows: very often (≥1 / 10); often (≥1 / 100, <1/10); infrequently (≥1 / 1000, <1/100); rarely (≥1 / 10,000, <1/1000); very rarely (<1/10 000) and unknown (impossible to estimate based on available data).
Immune system disorders | Infrequently - urticaria, skin rash, skin rashes |
Very rarely - Anaphylactic reactions * | |
Metabolic and nutritional disorders | Very often - hypoglycemia * |
Nervous system disorders | Rarely - peripheral neuropathy (acute pain neuropathy) |
Violations by the organ of vision | Infrequently - violations of refraction |
Infrequently - diabetic retinopathy | |
Violations of the skin and subcutaneous tissues | Infrequently - lipidodystrophy * |
General disorders and disorders at the site of administration | Infrequently - reactions at the injection sites, edema |
*Cm. Descriptions of individual adverse reactions
Descriptions of individual adverse reactions
Anaphylactic reactions. Very rare reactions of generalized hypersensitivity (including generalized skin rash, itching, increased sweating, gastrointestinal disorders, angioedema, breathing difficulties, rapid heartbeat, decreased blood pressure), which are potentially life-threatening, have been noted.
Hypoglycemia. Hypoglycemia is the most common side effect.It may develop if the insulin dose is too high in relation to the need for insulin. Severe hypoglycemia can lead to loss of consciousness and / or convulsions, temporary or irreversible dysfunction of the brain, or even death. Symptoms of hypoglycemia, as a rule, develop suddenly. They may include cold sweat, pale skin, fatigue, nervousness or tremor, anxiety, unusual tiredness or weakness, disorientation, decreased concentration, drowsiness, pronounced hunger, blurred vision, headache, nausea and palpitations. Clinical studies have shown that the incidence of hypoglycemia varies depending on the patient population, dosing regimen and glycemic control. In clinical studies, no differences were found in the overall incidence of episodes of hypoglycemia between patients receiving aspart insulin therapy and patients using human insulin preparations.
Lipodystrophy. It was reported infrequent cases of lipodystrophy. Lipodystrophy may develop at the injection site.
Contraindications
- increased individual sensitivity to insulin aspart or any of the components of the drug.
Do not use the drug NovoRapid® Penfill® in children under 2 years, because No clinical studies have been conducted in children under 2 years of age.
Use during pregnancy and lactation
NovoRapid® Penfill® can be prescribed during pregnancy. The data of two randomized controlled clinical trials (157 + 14 examined pregnant women) did not reveal any adverse effects of insulin aspart on the course of pregnancy or the health of the fetus / newborn compared to human insulin.
Careful monitoring of blood glucose levels and monitoring of pregnant women with diabetes mellitus (type 1 diabetes, type 2 diabetes or gestational diabetes) is recommended throughout pregnancy and during the period when pregnancy is possible. The need for insulin, as a rule, decreases in the first trimester and gradually increases in the second and third trimesters of pregnancy. Shortly after giving birth, the need for insulin quickly returns to the level that was before pregnancy.
In the period of breastfeeding NovoRapid® Penfill® can be used because administering insulin to a nursing mother does not pose a threat to the baby. However, it may be necessary to adjust the dose of the drug.
Application for violations of the liver
As with the use of other insulin preparations, patients with hepatic insufficiency should be more closely control the concentration of glucose in the blood and adjust the dose of insulin aspart individually.
Application for violations of kidney function
As with the use of other insulin preparations, patients with renal failure should be more closely control the concentration of glucose in the blood and adjust the dose of insulin aspart individually.
Use in children
Do not use the drug NovoRapid® Penfill® in children under 2 years, because No clinical studies have been conducted in children under 2 years of age.
Apply NovoRapid® Penfill® instead of soluble human insulin in children, it is preferable when a rapid onset of action of the drug is necessary, for example, when it is difficult for the child to observe the necessary time interval between injection and food intake.
Use in elderly patients
As with the use of other insulin preparations, elderly patients should be more closely control the concentration of glucose in the blood and adjust the dose of insulin aspart individually.
special instructions
Before a long trip associated with a change of time zones, the patient should consult with your doctor, because changing the time zone means that the patient must eat and inject insulin at another time.
Hyperglycemia
An inadequate dose of the drug or discontinuation of treatment, especially for type 1 diabetes mellitus, can lead to the development of hyperglycemia and diabetic ketoacidosis. As a rule, the symptoms of hyperglycemia appear gradually, over several hours or days. Symptoms of hyperglycemia include nausea, vomiting, drowsiness, redness and dryness of the skin, dry mouth, increased urine, thirst and loss of appetite, and the smell of acetone in exhaled air.Without appropriate treatment, hyperglycemia can lead to death.
Hypoglycemia
Skipping meals, unplanned increased physical exertion, or insulin dose that is too high relative to the needs of the patient can lead to hypoglycemia. After compensation of carbohydrate metabolism, for example, in case of intensified insulin therapy, the typical symptoms of hypoglycemia, which are typical for them, may change in patients, about which patients should be informed. The usual symptoms, precursors can disappear with a long course of diabetes.
The consequence of the pharmacodynamic features of the short-acting insulin analogues is that the development of hypoglycemia in their use may begin earlier than in the use of soluble human insulin.
Since NovoRapid® Penfill® should be used in direct connection with food intake, it is necessary to take into account the high rate of onset of the effect of the drug in the treatment of patients with comorbidities or receiving drugs that slow down the absorption of food. Concomitant diseases, especially infectious and accompanied by fever, usually increase the body's need for insulin. Dose adjustment of the drug may also be required if the patient has concomitant diseases of the kidneys, liver, dysfunction of the adrenal glands, pituitary, or thyroid gland.
When transferring a patient to other types of insulin,early symptoms, precursors of hypoglycemia may become less pronounced compared with those of the previous type of insulin.
Transferring a patient from other insulin preparations
Transferring a patient to a new type of insulin or another manufacturer’s insulin should be carried out under strict medical supervision. When changing the concentration, type, manufacturer and type (human insulin, animal insulin, human insulin analogue) of insulin preparations and / or method of manufacture, it may be necessary to change the dose or increase the frequency of injections compared to previously used insulin preparations. If necessary, dose adjustment, it can be made already at the first injection of the drug or during the first weeks or months of treatment.
Reactions at the injection site
As with the treatment of other insulin preparations, reactions can develop at the injection site, which manifests itself as pain, redness, urticaria, inflammation, hematoma, swelling and itching. Regularly changing the injection site in the same anatomical area can reduce symptoms or prevent the development of reactions. In very rare cases, it may be necessary to cancel NovoRapid® Penfill®.
The simultaneous use of preparations of the thiazolidinedione group and insulin preparations
Cases of the development of chronic heart failure have been reported in the treatment of patients with thiazolidinediones in combination with insulin preparations, especially when these patients have risk factors for the development of chronic heart failure.This fact should be taken into account when prescribing thiazolidinediones and insulin combination therapy to patients. When prescribing such a combination therapy, it is necessary to conduct medical examinations of patients in order to identify their signs and symptoms of chronic heart failure, weight gain and the presence of edema. If the symptoms of heart failure worsen in patients, treatment with thiazolidinediones should be discontinued.
Influence on ability to drive motor transport and control mechanisms
The ability of patients to concentrate and the speed of reaction may be impaired during hypoglycemia, which can be dangerous in those situations when these abilities are especially necessary (for example, when driving a vehicle or working with machines and mechanisms). Patients should be advised to take measures to prevent the development of hypoglycemia when driving and working with mechanisms. This is especially important for patients with the absence or decrease in the severity of symptoms, precursors of developing hypoglycemia or suffering from frequent episodes of hypoglycemia.
Overdose
Symptoms: a certain dose required for insulin overdose has not been established, however hypoglycemia may develop gradually if too high doses are administered in relation to the needs of the patient.
Treatment: mild hypoglycemia the patient can eliminate himself by ingesting glucose or sugar-containing foods. Therefore, patients with diabetes are advised to constantly carry with them sugar-containing products.
When severe hypoglycemia, when the patient is unconscious, you should enter from 0.5 mg to 1 mg of glucagon intramuscularly or n / a (a trained person can enter), or into / into a solution of glucose (dextrose) (only a medical professional can enter). It is also necessary to introduce dextrose in / in the case if the patient does not regain consciousness 10-15 minutes after the introduction of glucagon. After the recovery of consciousness, the patient is recommended to take carbohydrate-rich food to prevent the recurrence of hypoglycemia.
Drug interaction
There are a number of drugs that affect the need for insulin.
Hypoglycemic effect
The hypoglycemic effect of insulin weakens oral contraceptives, GCS, thyroid hormones, thiazide diuretics, Heparin, tricyclic antidepressants, sympathomimetics, somatropin, danazol, clonidine, Calcium channel blockers, diazoxide, morphine, phenytoin, nicotine.
Beta-blockers may mask the symptoms of hypoglycemia.
Octreotide / Lanreotide can both increase and decrease the need for insulin.
Alcohol can both enhance and reduce the hypoglycemic effect of insulin.
Incompatibility. Some drugs, such as those containing the thiol or sulfite groups, when added to the drug NovoRapid® Penfill® may cause the destruction of insulin aspart. Drug NovoRapid® Penfill® do not mix with other medicines. The exceptions are isophane-insulin and solutions for infusion.
Pharmacy sales terms
The drug is available on prescription.
Terms and conditions of storage
Store at 2 ° to 8 ° C (in the refrigerator), but not near the freezer. Do not freeze. Store cartridges in a carton box to protect from light. Should be protected from exposure to excessive heat and light. Opened cartridges should not be stored in the refrigerator. Store at a temperature not exceeding 30 ° C. Use within 4 weeks. Keep out of the reach of children.